Advances in next-generation sequencing (NGS) technologies have revolutionised the field of genomics, leading to an acceleration in data acquisition and a significant reduction of sequencing costs. Today, there are different sequencing approaches used for research and clinical applications.
However, with differing sequencing techniques, the optimal and most cost-effective technique for a particular application may not seem obvious.
This blog summarises the benefits and differentiating factors of the three most commonly used NGS technologies; targeted gene panels, whole-exome sequencing (WES) and whole-genome sequencing (WGS).
Targeted gene panels are often used as first-tier testing when a known gene or list of gene mutations may be the cause of the disease. This sequencing approach is favourable for its low cost, short turnaround time (TAT), and low rate of unspecific or incidental findings. Additionally, targeted gene panels have a higher depth of coverage than the two other sequencing technologies and, hence, are more likely to identify low-frequency variants in hot-spot genes. Targeted sequencing also produces less data, thus reducing analysis burden and data storage needs.
A gene panel often has better reimbursement than WES. However, occasionally payors believe that it can be challenging for clinicians to identify the underlying genetic cause of a disease and, hence, tend to run multiple genes panels before reaching a diagnosis. Therefore, these payors might approve WES or WGS to eliminate the need and cost of a series of panels.
WES provides a comprehensive analysis of the coding region (i.e., exon). While the exome, which comprises all exons, constitutes only ~1% of the whole genome, 85% of all disease-causing mutations are located in these regions.
WES is typically used when the clinician is uncertain of what gene or genes to look for or as second-tier test when a targeted gene panel assay could not identify a pathogenic mutation.
Unlike a targeted panel, WES allows for the identification of novel mutations. However, it may also produce a large number of variants of unknown significance (VUS). At the moment, WES is more expensive than targeted gene panel testing. It also has a relatively higher TAT and produces more data resulting in additional laboratory and data storage costs.
WGS is typically used as third-tier testing when targeted, and exome sequences have failed to determine the underlying pathogenic mutation of a disease. Unlike targeted and WES, WGS includes all intronic sequences, and can assist with identification of non-coding pathogenic variation, providing a more comprehensive view of exome coverage. WGS is the most expensive of these three techniques and requires higher TAT and data storage needs, and may produce a larger number of VUS.
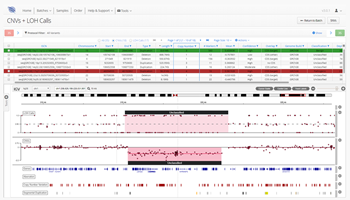
As one of the first steps in many NGS data analysis pipelines, accurate variant calling is often critical to downstream analysis and interpretation. Here, we take a look at variant calling best practice through a modern lens.
Read
A high-quality sequencing library is the linchpin to generating good sequencing data. We discuss our six top tips to help you improve your sequencing library.
Read
We discuss the development and current state of sequencing technologies, and where the future of NGS may take us...
Read
FISH is a cytogenetic technique utilised in labs to detect chromosomal abnormalities in both cancer and constitutional specimens. In this blog learn about the advantages of FISH...
Read
This blog will discuss FLT3’s normal function, its implications in myeloid malignancies, and the role of NGS in genetic identification and disease management of patients with FLT3 genetic alterations.
Read
While liquid biopsy may present an attractive alternative to a solid biopsy, it also has limitations. Here, we shed light on some advantages, limitations, and future outlook for liquid biopsy in oncology clinical practice.
Read
Next generation sequencing (NGS) is now in routine use for a broad range of research and clinical applications. Facilitating the detection of a wide variety of mutations, focus has never been higher on the value of making the correct choice for the initial sequence enrichment step, which, if poorly designed, can be a source of bias and error in the downstream sequencing assay.
Read