 Detecting cytogenetic abnormalities associated with multiple myeloma from bone marrow biopsies can be a tricky business. This is due to the fact that affected plasma cells make up only a small percentage of the sample, and grow poorly in cell culture. FISH is the gold standard method for detection and has been shown to play an important role in prognostic assessment. However, to further increase sensitivity, international guidelines recommend a CD138+ enrichment step upfront of performing FISH analysis.
Detecting cytogenetic abnormalities associated with multiple myeloma from bone marrow biopsies can be a tricky business. This is due to the fact that affected plasma cells make up only a small percentage of the sample, and grow poorly in cell culture. FISH is the gold standard method for detection and has been shown to play an important role in prognostic assessment. However, to further increase sensitivity, international guidelines recommend a CD138+ enrichment step upfront of performing FISH analysis.
We spoke to Theresa Brown, Director of the Cytogenetics Laboratory at Tulane University (New Orleans, Louisiana), about how her lab’s FISH analysis with OGT’s CytoCell® FISH probes benefited from using STEMCELL Technologies’ EasySep™ Human CD138 Positive Selection Kit and RoboSep™ Automated Cell Isolation Instrument after struggling with other CD138+ selection methods. She explains the improvements to her laboratory’s cell isolation process and the quality of the FISH results, increasing abnormality rates and enabling the laboratory to save time.
I’ve been doing this for a long time! I started in cytogenetics in 1990 as a technician in a bone marrow lab where I fell in love with it. This led me to go back to graduate school where I achieved my PhD in medical genetics, and I’ve been passionate and happy ever since! I have worked in a large cancer cytogenetics reference lab and then spent several years starting up labs, growing them and helping friends out, before I landed here at Tulane five years ago. We’re a small academic lab so we do a little bit of everything, but I would say that most of our work is haematology and oncology – along with some teaching and a little research.
We have a bone marrow transplant unit across the street at the hospital, so most of our specimens are from patients with myeloid disorders or multiple myeloma. A lot of our patients receive autologous stem cell transplants for myeloma and allogeneic transplants for myeloid disorders. 99% of our work is FISH and chromosome analysis on cancer specimens and we process around 15-25 multiple myeloma samples a month.
Improving patient care is of paramount importance for me, and we’re constantly performing quality checks on our work. We react to clinician requests and continually monitor updated guidelines, like the ones from WHO, to update our panels or add in the very latest new probes, as required. This ensures that our lab can offer the tests that we know the clinicians will be requesting. When I arrived at Tulane, most of our probes were already from CytoCell (with the exception of a few from another vendor that I didn’t particularly care for) and, thankfully, OGT had the rest of probes I needed. I am always looking for the best set of probes for our set up. Currently, we have a multiple myeloma FISH panel of six CytoCell probes – CKS1B/CDKN2C, MYC Breakapart, 9cen and 11cen for hyperdiploidy, IGH breakapart with reflex to the translocation partners if positive (including CCND1, FGFR3, MAF, MAFB, CCND3, and MYC), TP53 and 13q. I decided to use the CytoCell IGH breakapart probe for the first line analysis and then reflex to the partners because IGH analysis has always been problematic because of how it recombines. By using this algorithm and utilising the experience my technicians and I have gleaned over the years, we have pretty much ironed out all the issues we had been previously encountering with IGH.
Nowadays, I enjoy performing the IGH analysis and it makes me feel warm and fuzzy!
I really like CytoCell FISH probes, I’ve always had a good relationship with the company, in particular I appreciate the help and support that I receive. In the past, if any issue arose, I was usually told by other vendors that I was doing something wrong, but—when you’ve been doing FISH for 20 years—you know that you’re usually not doing something wrong!
OGT has also come in and helped me train my technicians, optimised procedures and offered a lot of help with our FFPE samples, as we don’t run many here. With that assistance, we can get them in and out very quickly. Ashley, our OGT FISH Field Application Scientist, has always made herself available to support our lab, whenever we have required assistance. As a result, I have stopped using all other probes and am strictly 100% CytoCell - I’m very happy!
Flow cytometry can pick up the CD138 and CD38 positive cells, but while it indicates that you have those positive cells, it doesn’t tell you which genetic mutations the patient is carrying. The NCCN guidelines outline the prognostic significance of genetic abnormalities identifiable by FISH, so this is something one must be very aware of as there are new drugs emerging for treatment. For example, there are different monoclonal antibodies that are inhibitors to specific genetic abnormalities. So, flow cytometry is only one part of the picture. It is very important, but it doesn’t give you the genetic part.
We’re lucky that our specimens are very fresh and we only need to walk over to the hospital to pick them up. As soon as we get them, we typically take about 1 ml of sample and immediately go to a cell isolation step. Then we will take the rest of the sample and set up two cultures, an unstimulated culture and a mitogen stimulated culture that grows for 96 hours. Finding cytogenetic abnormalities in the cell cultures can be difficult because plasma cells don’t typically divide in 24 hours; however, using a 96-hour stimulated culture increases the abnormality rates seen in the karyotypes.
From my experience, I feel that I get a better preparation. I think we’ve all experienced that the plasma cells can be sticky on the slide, but when selected with EasySep™ they just seem to spread better, making easier for the FISH analysis. They look better too - before I was having a really hard time - my techs didn’t like the previous method we were using, and I didn’t really like it. The FISH signals were very tough to read. In comparison, analysis on CD138+ enriched samples are much easier - we like reading these! There is no background, and you get much sharper FISH signals. The only problem we have is if there’s a very low percentage of cells, then we may have to do a lot of searching, but that’s not common.
Automating the enrichment is also very convenient; we can load it, walk away, do FISH setup, do analysis—do whatever we need to do—and then go back and finish it up.
Another advantage with automatic isolation, is that we tend to get a larger pellet size. So typically, we’re able to do the whole panel of 6 probes which means we get a better set of results. With a manual isolation, we were not always able to do a whole panel. Automation fits into the workflow well and I feel that my FISH results are equivalent, if not better than the flow results. In terms of abnormality rates, now, my abnormality rates are usually anywhere from 40 to 50%. I feel confident because I know that we’re finding the abnormalities, we’ve got the right cells and we’re getting good results.
It’s been a perfect marriage! I like OGT and I also like STEMCELL Technologies. Both workflows are very straightforward, very easy - I get consistent results. That’s what I want - consistent results for my patients, and I have that. I woke up a few months ago and realised that I’m now an elder / mentor in this field and I have a lot of people from all over the world asking for my help, opinion, advice and my SOPs. I don’t mind because I love teaching people who want to learn about genetics and cytogenetics. I constantly recommend OGT’s CytoCell probes and STEMCELL Technologies platform because they work. If you need help, you can get it from both companies. For example, I’ve sometimes had a surprising FISH result, I know I can always just send out an email to OGT.
Laboratories must undertake all appropriate validation of any LDT as per 42 CFR 493.1253 - Standard: Establishment and verification of performance specifications.
The tests discussed in this report were developed and their performance characteristics determined by the Hayward Genetics Center, Tulane University Medical School. They have not been cleared or approved by the U.S. Food and Drug Administration.

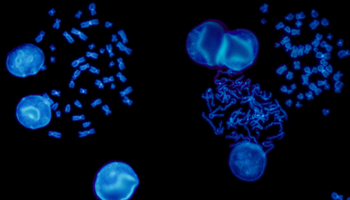
FISH is a cytogenetic technique utilised in labs to detect chromosomal abnormalities in both cancer and constitutional specimens. In this blog learn about the advantages of FISH...
Read
Carmen Vokaty, Assistant-Chief Medical Technologist in Cytogenetics at the McGill University Health Centre, details how her lab incorporated OGT’s Cytocell® Tissue Pretreatment kit into its workflow after struggling with a homebrew solution.
Read
Dr Jennie Thurston, Director of Cytogenetics, Carolinas Pathology Group, Atrium Health, explains how OGT's CytoCell® FISH probes were seamlessly incorporated into the automated workflow for fast, high quality results.
Read
Dr. Theresa C. Brown, Tulane University School of Medicine, answer questions around her AGT 2017 presentation 'FISHing with the real-life laboratory experts.'
Read
Megumi Hada, PhD, Senior Research Scientist, RaISE, Prairie View, Texas A&M. answers our questions on space radiation, how it affects astronaut health, and what her research can teach us about the biological effects of radiation here on Earth.
Read